Innovative technologies that streamline
workflow, boost
productivity, and
promote
patient-centric care
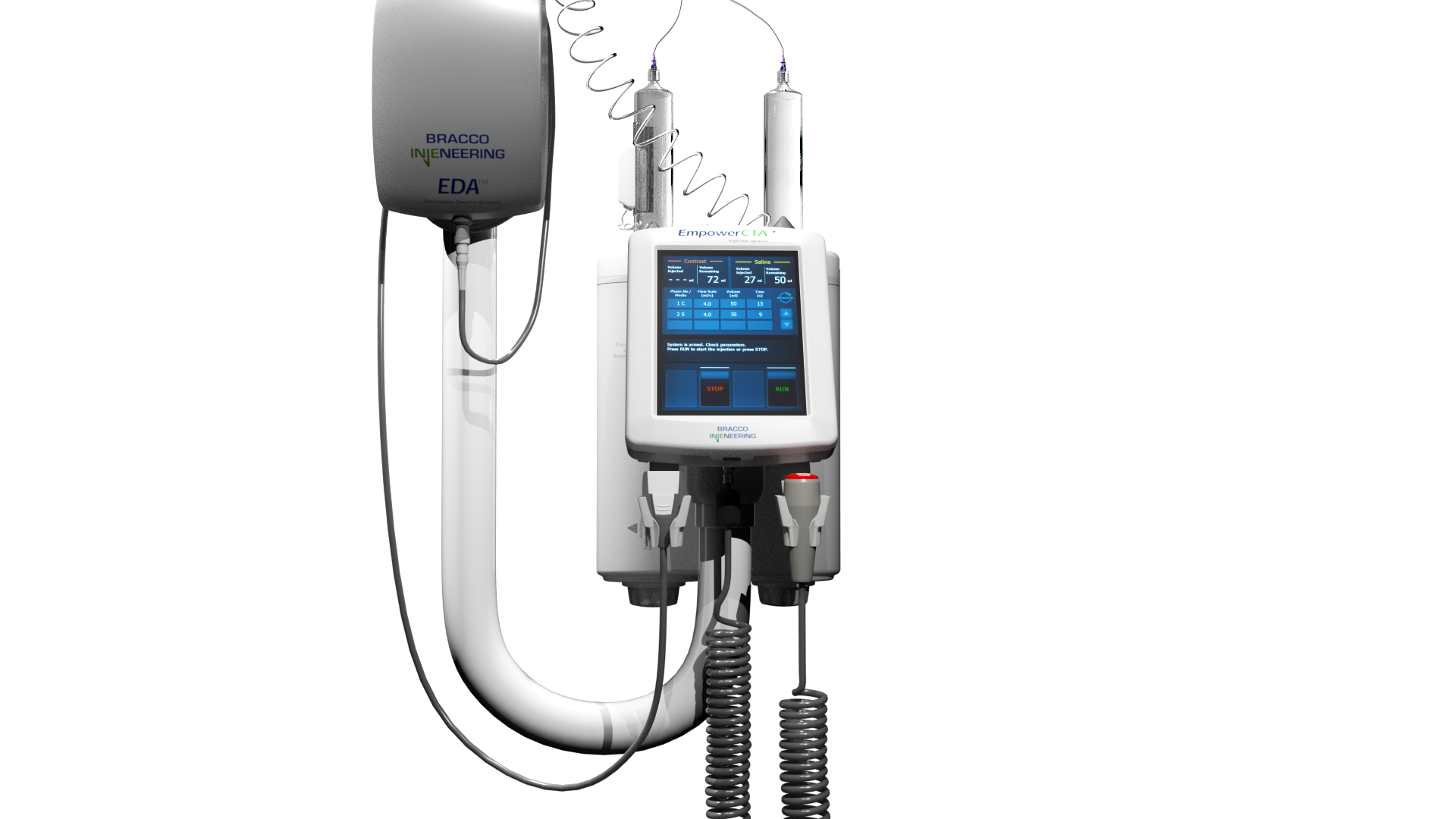
EmpowerCTA®+ Injector System
INDICATIONS:
The EmpowerCTA®+ Injector System is indicated for the vascular administration of
contrast and
flushing
media in conjunction with computed tomography (CT) scanning of the body with an
optional interface to a CT scanner and an optional calculator for glomerular filtration rate
(GFR).
The Extravasation Detection Accessory (EDATM) is an optional accessory and is
indicated for
the
detection of extravasations of
ionic and nonionic contrast during CT procedures using a power injector.
CONTRAINDICATIONS:
The EmpowerCTA+ Injector System is not intended for use as a long-term infusion pump, nor is
it intended
to be used to inject any agents other than
contrast or flushing media. Do not attempt to use the Injector for any other purpose (such
as
chemotherapy or drug infusion). The EmpowerCTA+ Injector System should
not be used to inject substances into nonvascular body cavities. Any applications of the
EmpowerCTA+
Injector System other than those described in its Operator Manual are inappropriate
and should not be attempted.
Protocols have been independently developed and are not intended as medical advice. Bracco
and Bracco
Injeneering S.A. shall not be responsible for any physicians’ reliance on these or
any other products.
Not all products available in all global markets.
EmpowerCTA+ is distributed by Bracco Diagnostics Inc., 259 Prospect Plains Road, Bldg. H,
Monroe
Township, NJ 08831 USA; Phone: (800) 631-5245; Fax: (609) 514-2424.
EmpowerCTA+ is manufactured by Bracco Injeneering S.A., Avenue de Sevelin 46, CH–1004
Lausanne,
Switzerland. https://imaging.bracco.com
Customer Service: 1-877-BRACCO 9 (1-877-272-2269)
Professional Services: 1-800-257-5181, Option 2
EmpowerCTA+ Email: empowerinfo@diag.bracco.com
About Bracco Injeneering S.A.
Bracco Injeneering S.A., located in Lausanne, Switzerland, is the most recently added
Business Unit of
Bracco Imaging. Bracco Injeneering is committed to develop the best-in-class integrated
injection solutions with a strong heritage in research and innovation. It provides quality
solutions for
state-of-the-art radiology centers, offering proven injection technology, built
on Bracco Imaging’s expertise.
Thanks to this strengthened product portfolio, including the CT Exprès® 3D Contrast Media Delivery System, EmpowerCTA+ Injector System, EmpowerMR® Injector System, NEXO®
Contrast Management System and NEXO [DOSE]® Multi-Modality Radiation Informatics,
Bracco
Imaging will be able to focus on constant innovation, not only for devices, but also for
software development and data management.
CT Exprès, EmpowerCTA, IRiSCT, EmpowerMR, EmpowerSync, NEXO and NEXO [DOSE] are registered
trademarks of
Bracco Injeneering S.A.
EDA is a trademark of Bracco Injeneering S.A.
NEXO® Contrast Management System
Intended Use:
NEXO® Contrast Management System is a server-based application intended to be used as a data-management and visualization system. NEXO Contrast Management System is able to read, store, share, compare/relate, as well as graphically display data and injection programs coming from RIS, PACS and multiple enabled EmpowerCTA® Injector Systems. It provides the users with record-lists, graphics and reports about data and performances.
This device is intended for retrospective and statistical data management and visualization. It is not intended for real time data visualization, diagnostic applications or for any other use for which the device is not indicated. This device is only to be operated by and under quasi-continuous supervision of qualified and trained staff in an appropriate licensed healthcare facility.
NEXO Contrast Management System is compatible with the EmpowerCTA® Injector System version 8.0, and the EmpowerCTA®+ Injector System version 9.0 and higher.
Bracco Injeneering S.A. reserves the right at any time and without notice, to change the specifications and features described herein, or to change the production or adjust the product described.
Not all products available in all global markets
NEXO is distributed by Bracco Diagnostics Inc. 259 Prospect Plains Road, Building H, Monroe Township, NJ 08831 USA; Phone: (800) 631-5245; Fax: (609) 514-2424.
NEXO Customer Service: 1-877-BRACCO 9 (1-877-272-2269)
NEXO questions: Nexo.Answers@diag.bracco.com
NEXO U.S. Technical Service and Support: Phone: 888-670-7701; Fax: 609-514-2448;
NEXO Email: customer.support@acistmedical.com
NEXO is manufactured by Bracco Injeneering S.A., Avenue de Sévelin 46, CH-1004 Lausanne, Switzerland. https://imaging.bracco.com
About Bracco Injeneering S.A.
Bracco Injeneering S.A., located in Lausanne, Switzerland, is the most recently added Business Unit of Bracco Imaging. Bracco Injeneering is committed to develop the best-in-class integrated injection solutions with a strong heritage in research and innovation. It provides quality solutions for state-of-the-art radiology centers, offering proven injection technology, built on Bracco Imaging’s expertise.
Thanks to this strengthened product portfolio, including the CT Exprès® 3D Contrast Media Delivery System, EmpowerCTA®+ Injector System, EmpowerMR® Injector System, NEXO Contrast Management System and NEXO [DOSE]® Multi-Modality Radiation Informatics, Bracco Imaging will be able to focus on constant innovation, not only for devices, but also for software development and data management.
CT Exprès, EmpowerCTA, EmpowerMR, NEXO and NEXO [DOSE] are registered trademarks of Bracco Injeneering S.A.
Go to: imaging.bracco.com/us-en
Bracco Diagnostics Inc.
259 Prospect Plains Road, Building H
Monroe Township, NJ 08831 USA
Phone: 609-514-2200
Toll Free: 1-800-631-5245 (U.S. only)
Fax: 609-514-2424


